The Alabama Academy of Eye Physicians and Surgeons is urging residents of the Yellowhammer State to practice caution when selecting eye drops.
As many as 68 people throughout the country have reported infections linked to the bacteria, Pseudomonas aeruginosa, after using contaminated eye drops, according to a news release. The infections have resulted in three deaths and six people having their eyes removed. Before the eye infections, Pseudomonas aeruginosa had never been documented in the U.S.
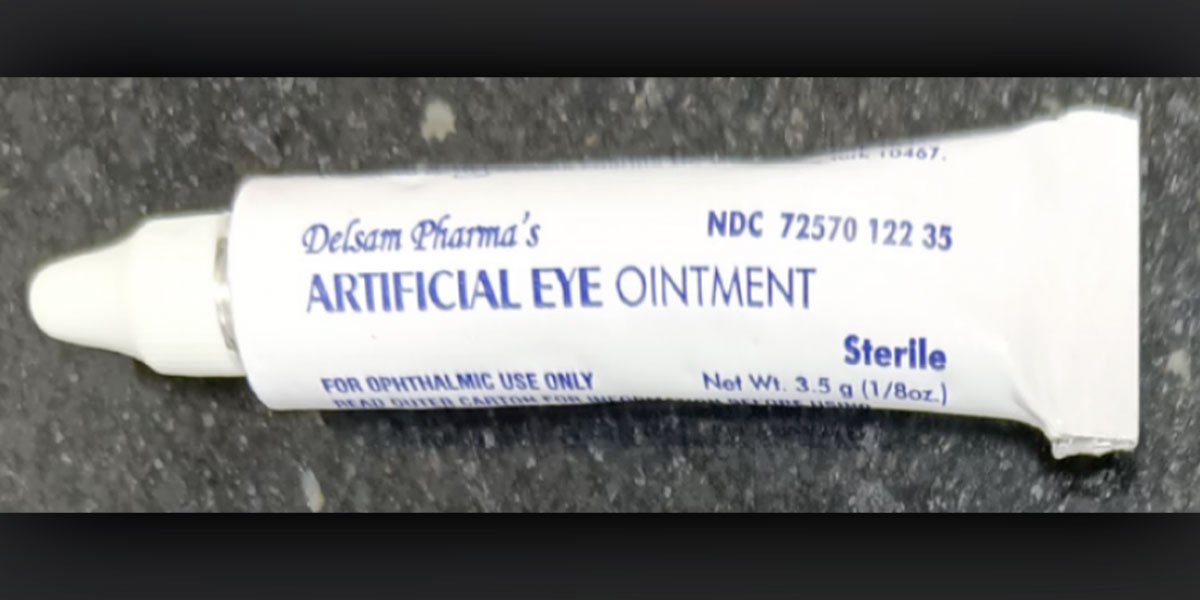
The eye drops, which are manufactured in India, that reportedly contain the bacteria are: Delsam Pharma’s Artificial Eye Ointment, EzriCare Artificial Tears, and Delsam Pharma’s Artificial Tears, the news release said.
If you used Ezricare artificial tears or other recalled products associated w/ Pseudomonas aeruginosa outbreak, please stop using and seek care immediately. Get the latest updates on this multi-state outbreak: https://t.co/tumUneiSv6 #AntimicrobialResistance pic.twitter.com/nmzJ9wbEfo
— CDC Antimicrobial Resistance (@CDC_AR) March 22, 2023
Medical professionals, including those in Alabama, have addressed the infections caused by the eye drops.
“Redness, pain and discharge are some of the early signs and symptoms of an eye problem, whether you’re using these products or not,” said Dr. Russell Read with UAB Callahan Eye Hospital. “If you have any of these symptoms or something out the ordinary, immediately contact your eye care provider. This is especially important if you wear contacts, which can make you more prone to infections.”
The Center for Disease Control and Prevention (CDC) said consumers should stop using the “extensively drug resistant” eye drops and seek medical attention as soon as possible.
“Patients and healthcare providers should immediately stop the use of EzriCare Artificial Tears,” the agency said. “Patients who have used EzriCare or Delsam Pharma’s artificial tears and who have signs or symptoms of an eye infection should seek medical care immediately.”
Austen Shipley is a staff writer for Yellowhammer News.